As with any decision you make about your healthcare, it is important to make an informed decision when choosing vein treatment. Safe, effective vein treatment options are more numerous than ever before, and we understand that sorting through the choices can be confusing. Our Board-Certified vein specialists are here to help guide you in this process and will advise you on the best options for your needs taking into account the condition of your veins, your overall health, and your goals for treatment. Read on to learn about three of the most successful vein treatments currently available, how they work, and who can benefit the most from each one.
VenaSealTM
VenaSealTM is a state-of-the-art technique that permanently closes off varicose veins. This means that VenaSealTM treats the underlying cause rather than merely acting as a temporary or purely cosmetic procedure. VenaSealTM is performed using local anesthesia applied only to the area being treated and works by injection of a medical grade adhesive into the affected vein(s). The adhesive causes a localized response in the wall of the vein that causes scar tissue to form, permanently sealing the vein[1].
VenaSealTM effectively eliminates or greatly reduces varicose vein symptoms and prevents future health complications that can arise from chronic venous hypertension[1].
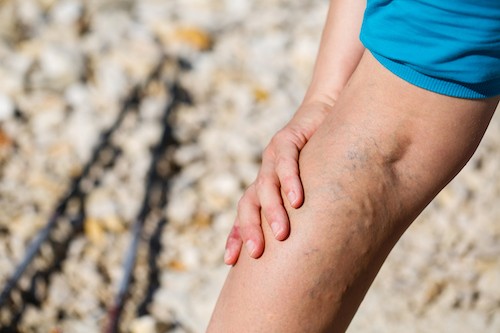
You may be a candidate for VenaSealTM if you are suffering from common varicose vein symptoms such as swelling, muscle fatigue, itching, tingling, and numbness. VenaSeal TM is also an excellent option for more advanced symptoms, such as inflammation within the veins (thrombophlebitis) or skin wounds (varicose ulcers).
In a comparison study of six different treatments VenaSealTM received the highest marks for anatomical success, meaning complete closure of the vein within 6 months, and for reduction of post treatment pain. Additionally, VenaSealTM has shorter healing times than other techniques, so you can return to work, sports, and recreational activities sooner, often the same day.
Desert Vein and Vascular Institute is proud to be the #1 provider of VenaSealTM in the USA, having performed over 19,000 VenaSealTM procedures.
ClosureFastTM
ClosureFastTM is a curative procedure for varicose veins that uses heat generated by radiofrequency (microwaves) to close off affected veins. ClosureFastTM is performed in-office using local anesthesia and was designed to decrease discomfort and complications associated with conventional surgical techniques in which varicose veins are tied off and then removed.
- This is a preferred technique for larger varicose veins and is associated with nearly 100% success for complete vein closure[3].
ClosureFastTM provides superior results including greater symptom improvement, faster recovery with less pain and bruising, faster return to work, and more improved quality of life compared to surgical options[3,4]. ClosureFastTM also takes significantly less time to perform, which means less local anesthesia is required[4].
Most ClosureFastTM patients are able to return to work in less than 5 days, compared to an average of 12 days with conventional treatments.
Sclerotherapy
In this procedure a solution; either a liquid, such as a salt, or a foam compound, is injected into a varicose vein causing the vein to scar and eventually collapse. Blood then reroutes into healthy veins. Over a few weeks’ time the collapsed veins are reabsorbed into the surrounding tissue. Sclerotherapy is often the preferred treatment for smaller varicose veins and spider veins that are close to the surface and is also used for pelvic venous insufficiency and venous malformation[5].
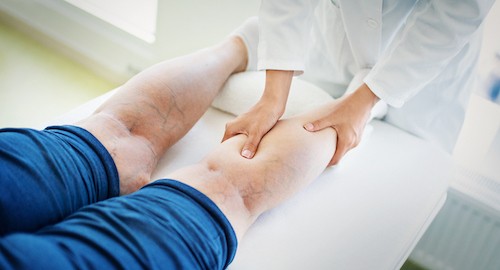
Overall, patients report less pain and bruising with foam sclerotherapy compared to conventional surgery. In one study, 43% of participants were able to return to work within 24 hours compared to none of the surgical patients[6].
An important thing to know when considering sclerotherapy is that the type of compound used in the procedure makes a big difference in the results. At Desert Vein and Vascular Institute, we use a medication called Asclera® exclusively. Asclera® has been proven safe and effective in clinical trials and is the only FDA-approved sclerosing compound on the market. Superior manufacturing and quality control standards ensure that Asclera® provides unsurpassed purity and guaranteed levels of active ingredient, providing consistently excellent and reliable results.
Visible improvements with sclerotherapy are usually gradual and this procedure can require several treatments to achieve the full, desired effects[5].
To learn about which vein treatment is right for you we invite you to schedule a free consultation at Desert Vein and Vascular Institute. All of our physicians are board-certified vascular surgeons who specialize in helping people like you.
To schedule a free consultation, please call 1-800-827-4267 today.
References
- Expert review on the VenaSeal® system for endovenous cyano-acrylate adhesive ablation of incompetent saphenous trunks in patients with varicose veins. Expert Rev Med Devices, 2017. 14(10): p. 755-762
https://pubmed.ncbi.nlm.nih.gov/28892412/ - Network meta-analysis to compare VenaSeal with other superficial venous therapies for chronic venous insufficiency. J Vasc Surg Venous Lymphat Disord, 2020. 8(3): p. 472-481.e3
https://pubmed.ncbi.nlm.nih.gov/32063522/ - Outcome of ClosureFAST radiofrequency ablation for large-diameter incompetent great saphenous vein. Ann Surg Treat Res, 2019. 96(6): p. 313-318
https://pubmed.ncbi.nlm.nih.gov/31183336/ - Three-year follow-up and quality of life of endovenous radiofrequency ablation of the great saphenous vein with the ClosureFast™ procedure: Influence of BMI and CEAP class. Vascular, 2018. 26(5): p. 498-508
https://pubmed.ncbi.nlm.nih.gov/29486654/ - Liquid and Foam Sclerotherapy for Spider and Varicose Veins. Surg Clin North Am, 2018. 98(2): p. 415-429
https://pubmed.ncbi.nlm.nih.gov/29502781/ - Foam Sclerotherapy: An Emerging, Minimally Invasive And Safe Modality Of Treatment For Varicose Veins. J Ayub Med Coll Abbottabad, 2019. 31(Suppl 1)(4): p. S641-s645
https://pubmed.ncbi.nlm.nih.gov/31965766/ - Phlebectomy Techniques for Varicose Veins. Surg Clin North Am, 2018. 98(2): p. 401-414
- Evidence summary of combined saphenous ablation and treatment of varicosities versus staged phlebectomy. J Vasc Surg Venous Lymphat Disord, 2017. 5(1): p. 134-137
https://pubmed.ncbi.nlm.nih.gov/27987603/
